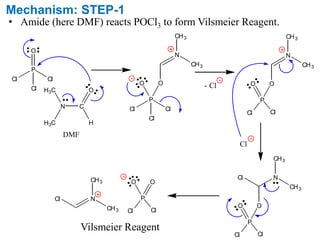
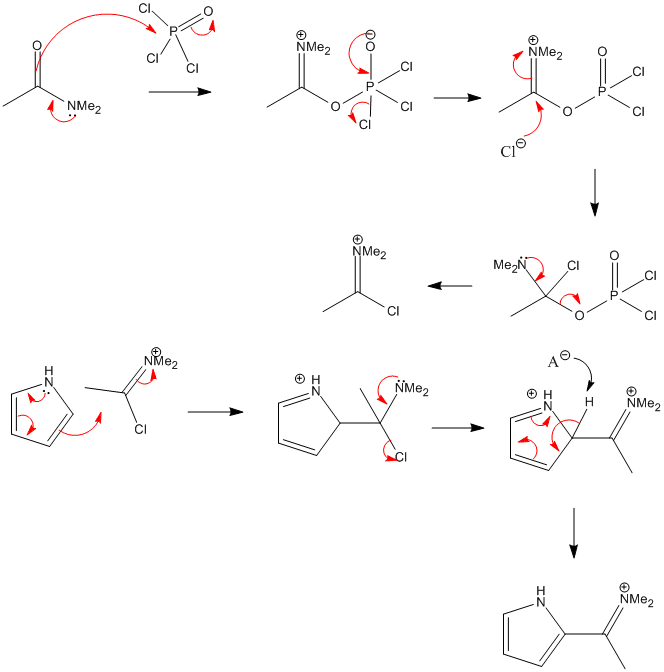
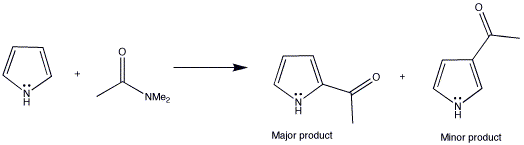
Advanced Organic Chemistry: Reaction Mechanism, Strategy, Applications. - The Vilsmeier–Haack reaction (also called the Vilsmeier reaction) is the chemical reaction of a substituted amide with phosphorus oxychloride and an electron-rich arene to

Vilsmeier–Haack reactions of carbonyl compounds: synthesis of substituted pyrones and pyridines - ScienceDirect
Synthesis of some new 1-aryl-4-formyl-3-(4-hydroxy-6-methyl-2-oxo -2H-pyran-3-yl)pyrazoles using the Vilsmeier-Haack reaction--isolation of the key intermediate 1-aryl-3-(4-hydroxy-6-methyl-2-oxo-2H-pyran-3-yl)pyrazoles - Document - Gale Academic OneFile

Scheme 1. Vilsmeier-Haack mechanism for formyl-de-hydrogenation of P3HT. | Download Scientific Diagram
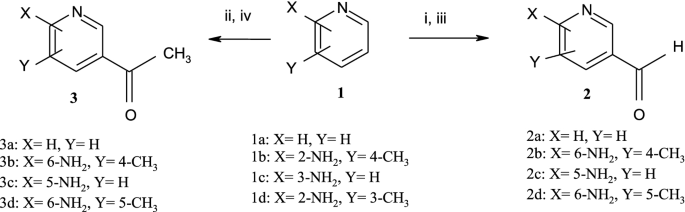
Micellar effects on kinetics and mechanism of Vilsmeier–Haack formylation and acetylation with Pyridines | SpringerLink
![Vilsmeier–Haack reaction of 7-acetyl-2-arylindoles: a convenient method for the synthesis of 6-oxo-6H-pyrrolo[3,2,1-ij]quinoline-1,5-dicarbaldehydes - Organic & Biomolecular Chemistry (RSC Publishing) Vilsmeier–Haack reaction of 7-acetyl-2-arylindoles: a convenient method for the synthesis of 6-oxo-6H-pyrrolo[3,2,1-ij]quinoline-1,5-dicarbaldehydes - Organic & Biomolecular Chemistry (RSC Publishing)](https://pubs.rsc.org/en/Content/Image/GA/C8OB03040E)