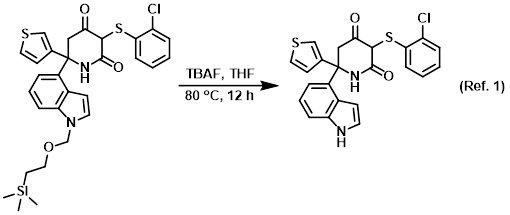
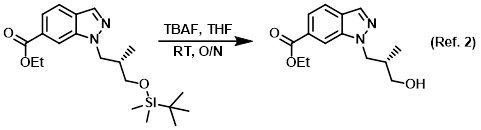
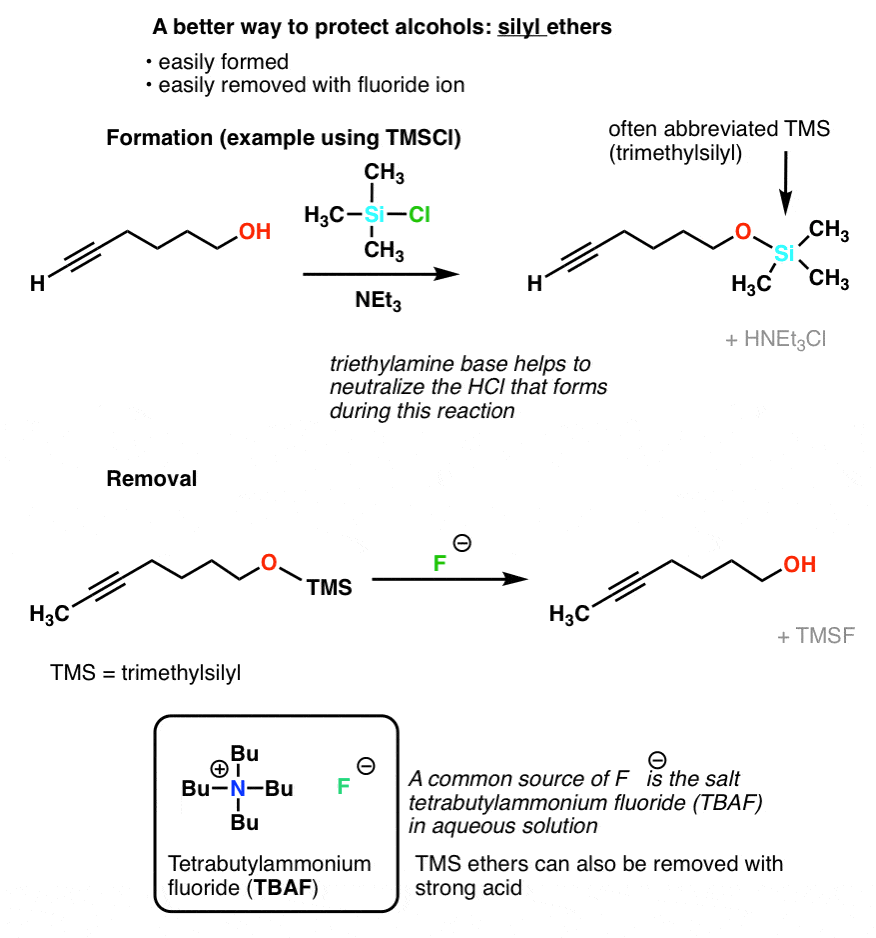
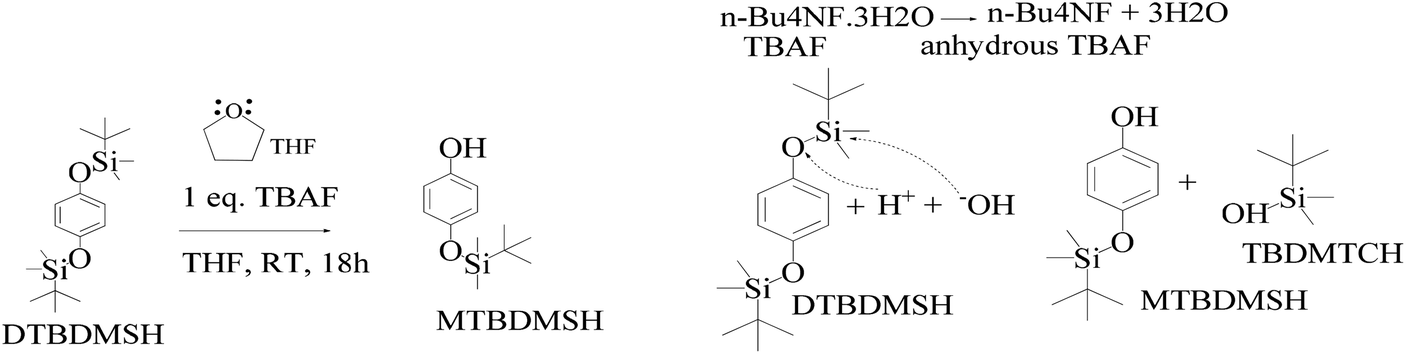
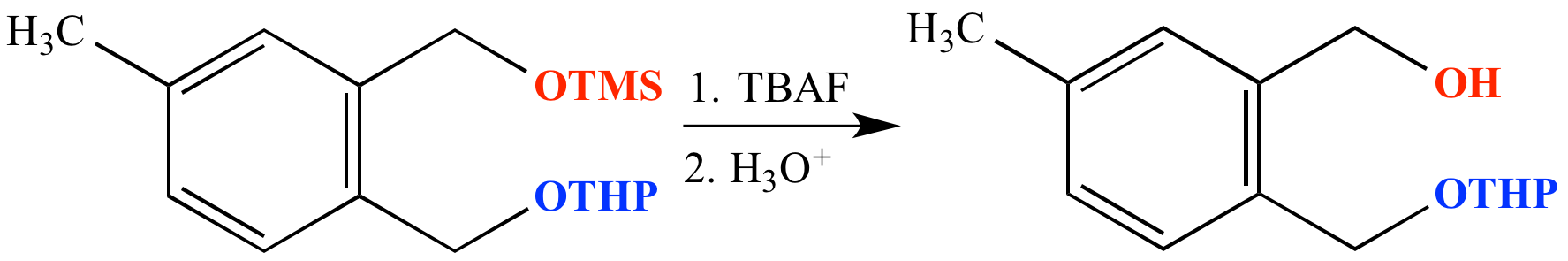SciELO - Brasil - An Efficient and Chemoselective Deprotection of Aryl <i>tert</i>-Butyldimethylsilyl (TBDMS) Ethers by NaCN An Efficient and Chemoselective Deprotection of Aryl <i>tert</i>-Butyldimethylsilyl (TBDMS) Ethers by NaCN
A new dendrimer series: synthesis, free radical scavenging and protein binding studies - RSC Advances (RSC Publishing) DOI:10.1039/D0RA04102E

Cyclization reactions of 2-alkynylbenzyl alcohol and 2-alkynylbenzylamine derivatives promoted by tetrabutylammonium fluoride - ScienceDirect
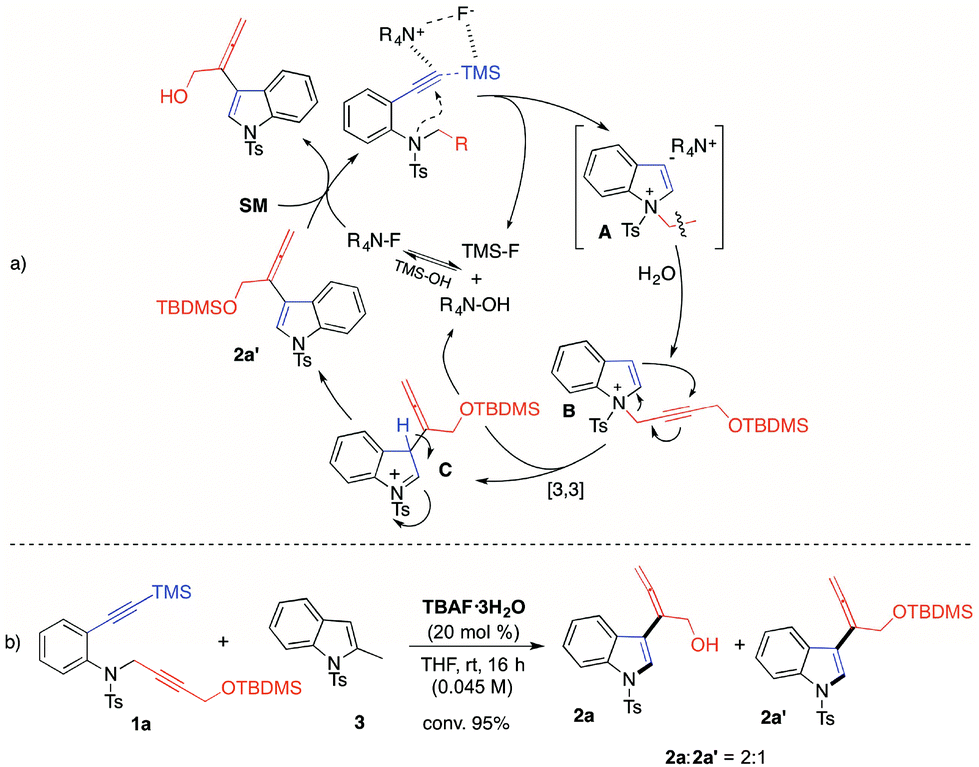
TBAF catalyzed one-pot synthesis of allenyl-indoles - Organic Chemistry Frontiers (RSC Publishing) DOI:10.1039/C7QO00414A

Alkyne Cycloadditions Mediated by Tetrabutylammonium Fluoride: A Unified and Diversifiable Route to Isoxazolines and Pyrazolines | Organic Letters

Regioselective O2′,O3′‐Deacetylations of Peracetylated Ribonucleosides by Using Tetra‐n‐butylammonium Fluoride - Babu Kumar - 2014 - European Journal of Organic Chemistry - Wiley Online Library
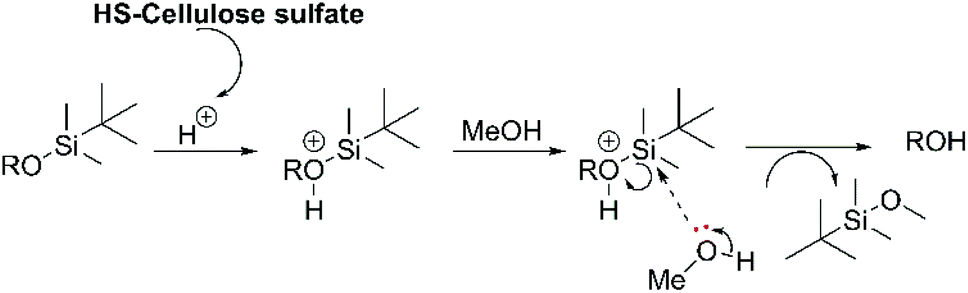
Highly sulphated cellulose: a versatile, reusable and selective desilylating agent for deprotection of alcoholic TBDMS ethers - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C8OB01438H
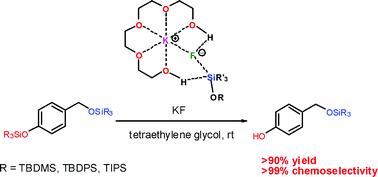
A mild and efficient method for the selective deprotection of silyl ethers using KF in the presence of tetraethylene glycol - Organic & Biomolecular Chemistry (RSC Publishing)