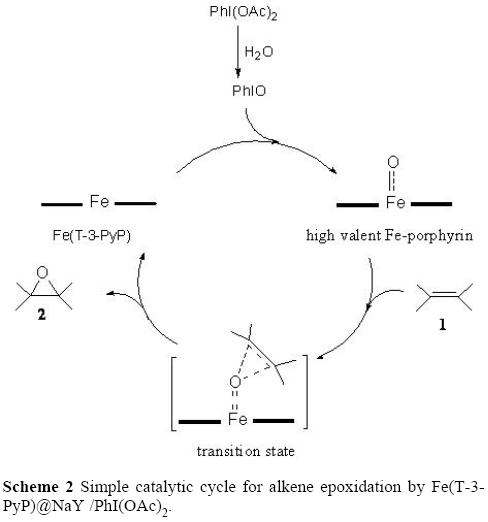
Catalytic hypervalent iodine oxidation of alcohols to corresponding aldehydes or ketones using 2,2,6,6-tetramethylpiperidinyl-1-oxy and potassium peroxodisulfate | SpringerLink
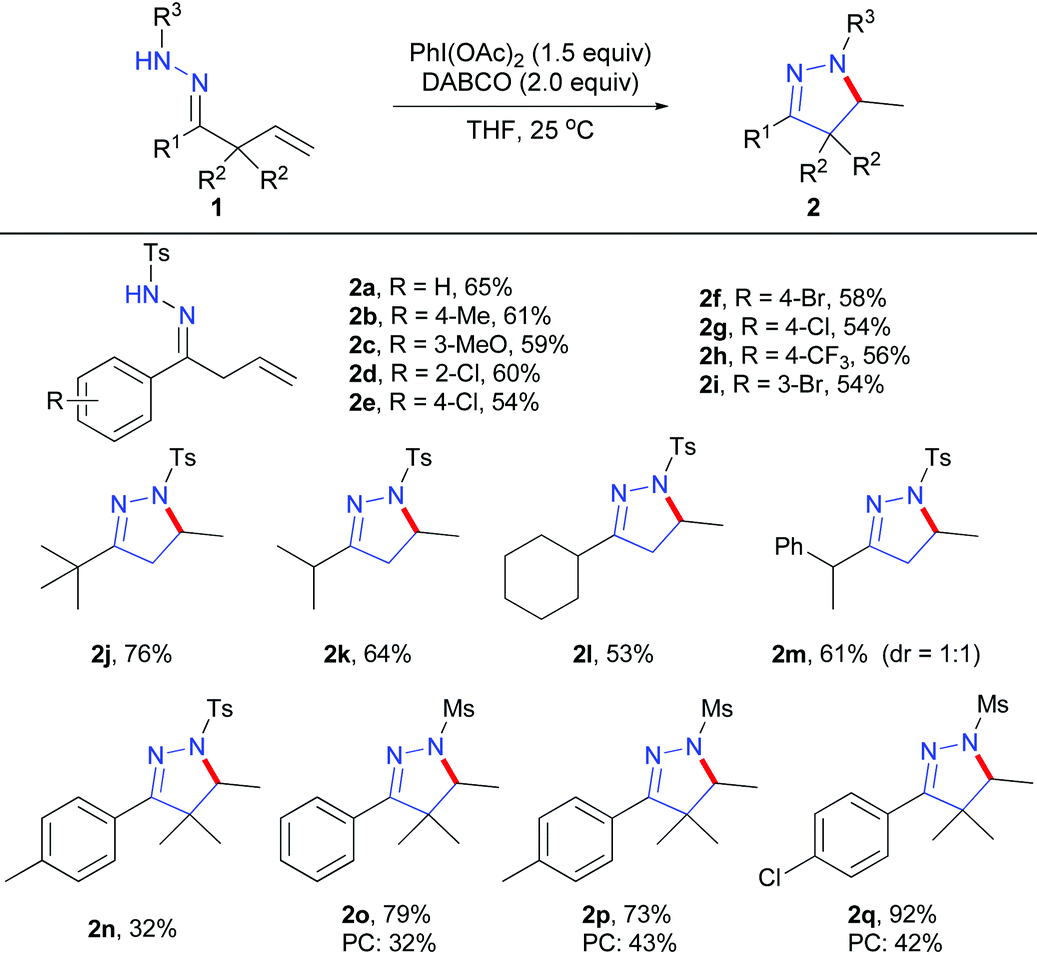
PhI(OAc)2-mediated functionalisation of unactivated alkenes for the synthesis of pyrazoline and isoxazoline derivatives - Organic & Biomolecular Chemistry (RSC Publishing)
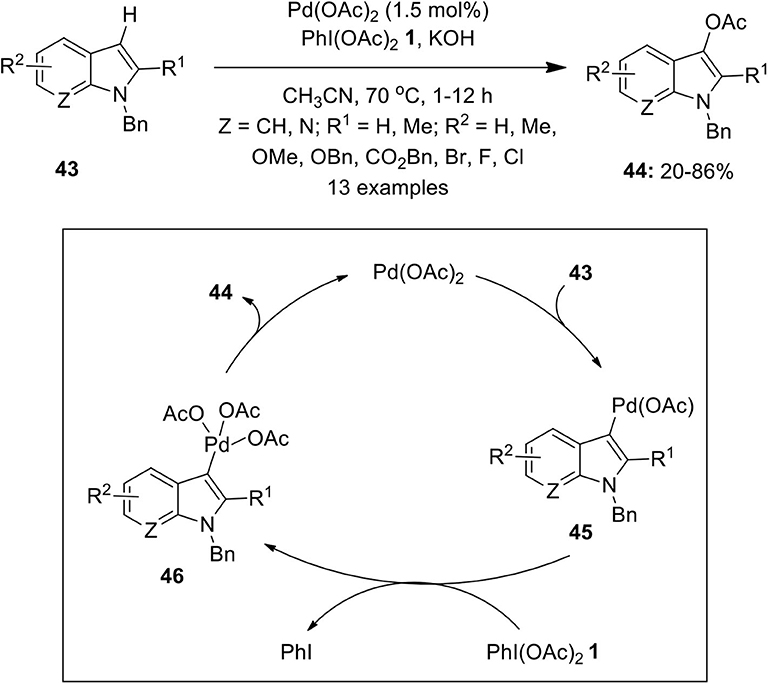
Frontiers | Hypervalent Iodine Reagents in Palladium-Catalyzed Oxidative Cross-Coupling Reactions | Chemistry

Copper-Promoted Functionalization of Organic Molecules: from Biologically Relevant Cu/O2 Model Systems to Organometallic Transformations. - Abstract - Europe PMC

Copper‐Catalyzed Decarboxylative Methylation of Aromatic Carboxylic Acids with PhI(OAc)2 - Jiang - 2014 - European Journal of Organic Chemistry - Wiley Online Library

Molecules | Free Full-Text | Hypervalent Iodine Reagents in High Valent Transition Metal Chemistry | HTML

Frontiers | Hypervalent Iodine-Mediated Diastereoselective α-Acetoxylation of Cyclic Ketones | Chemistry

PDF) Visible-Light, Iodine-Promoted Formation of N-Sulfonyl Imines and N-Alkylsulfonamides from Aldehydes and Hypervalent Iodine Reagents

The reaction of terminal alkynes with PhI(OAc)2: a convenient procedure for the preparation of α-acyloxy ketones - ScienceDirect

Mechanistic investigation into the C(sp3)–H acetoxylation of morpholinones - Chemical Science (RSC Publishing)

PhI(OAc)2-promoted metal-free oxidation of 2-oxoaldehydes: a facile one-pot synthesis of cyanoformamides - RSC Advances (RSC Publishing)

A highly efficient TEMPO mediated oxidation of sugar primary alcohols into uronic acids using 1-chloro-1,2-benziodoxol-3(1H)-one at room temperature - ScienceDirect

Synthesis of β-acetoxy alcohols by PhI(OAc)2-mediated metal-free diastereoselective β-acetoxylation of alcohols - Organic & Biomolecular Chemistry (RSC Publishing)