
Why does ring opening reaction (of lactones) often follow nucleophilic pathway and not electrophilic?
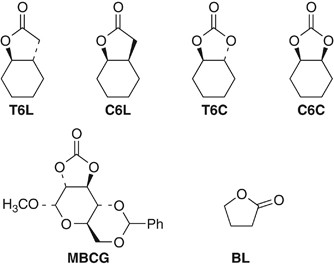
Ring-opening polymerization of a five-membered lactone trans-fused to a cyclohexane ring | Polymer Journal

W(OTf)6‐Catalyzed Synthesis of γ‐Lactones by Ring Contraction of Macrolides or Ring Closing of Terminal Hydroxyfatty Acids in Ionic Liquid - Xie - 2018 - ChemSusChem - Wiley Online Library

Ring-opening polymerization of γ-lactones and copolymerization with other cyclic monomers - ScienceDirect

Why does ring opening reaction (of lactones) often follow nucleophilic pathway and not electrophilic?

Ring opening polymerization of β-acetoxy-δ-methylvalerolactone, a triacetic acid lactone derivative - ScienceDirect

Ring-opening polymerization of a macrocyclic lactone monomer isolated from oligomeric byproducts of poly(butylene succinate) (PBS): An efficient route to high-molecular-weight PBS and block copolymers of PBS - ScienceDirect

Enzymatic Ring‐Opening Polymerization of Lactones: Traditional Approaches and Alternative Strategies - Engel - 2019 - ChemCatChem - Wiley Online Library

Synthetic schemes of poly(ester) by ring opening polymerization with... | Download Scientific Diagram
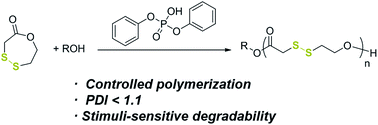
Synthesis of poly(disulfide)s with narrow molecular weight distributions via lactone ring-opening polymerization - Chemical Science (RSC Publishing)

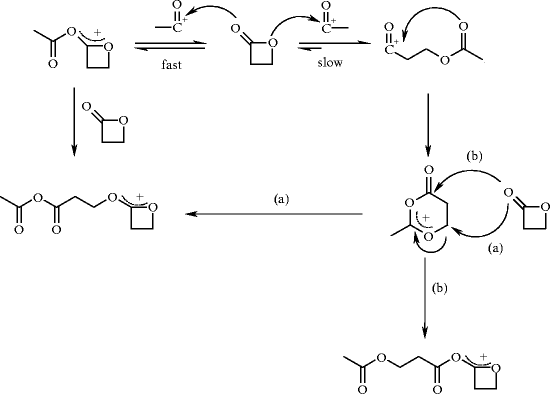
Scheme 8. Ring-opening of a -lactone upon either (a) an oxygenacyl or... | Download Scientific Diagram
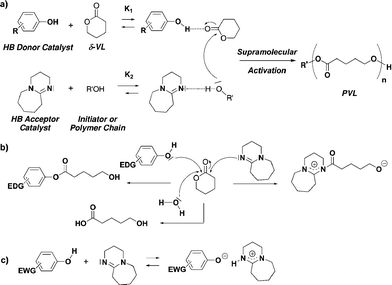
Ring-opening polymerization of lactones using supramolecular organocatalysts under simple conditions - RSC Advances (RSC Publishing) DOI:10.1039/C2RA22535B
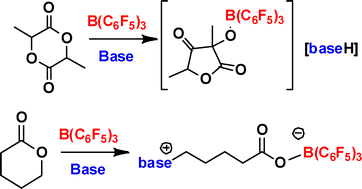
Ring openings of lactone and ring contractions of lactide by frustrated Lewis pairs - Dalton Transactions (RSC Publishing)

Ring-opening polymerization of γ-lactones and copolymerization with other cyclic monomers - ScienceDirect

Controlled Random Terpolymerization of β-Propiolactone, Epoxides, and CO2 via Regioselective Lactone Ring Opening | CCS Chem

Completely recyclable biopolymers with linear and cyclic topologies via ring -opening polymerization of γ-butyrolactone | Nature Chemistry
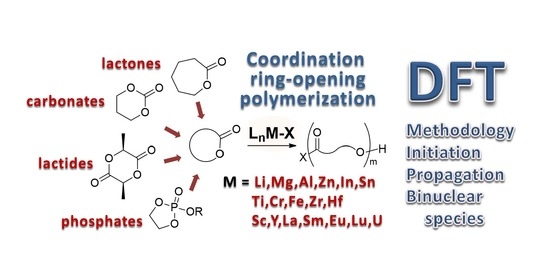
Molecules | Free Full-Text | Coordination Ring-Opening Polymerization of Cyclic Esters: A Critical Overview of DFT Modeling and Visualization of the Reaction Mechanisms



![ASAP] Lossen Rearrangement vs C–N Reductive Elimination Enabled by ASAP] Lossen Rearrangement vs C–N Reductive Elimination Enabled by](https://www.researcher-app.com/image/eyJ1cmkiOiJodHRwczovL3MzLWV1LXdlc3QtMS5hbWF6b25hd3MuY29tL3N0YWNrYWRlbWljL3Byb2R1Y3Rpb24vcGFwZXIvNTU0Mjg4OS5wbmciLCJmb3JtYXQiOiJ3ZWJwIiwicXVhbGl0eSI6MTAwLCJ3aWR0aCI6NTEyLCJub0NhY2hlIjp0cnVlfQ==.webp)