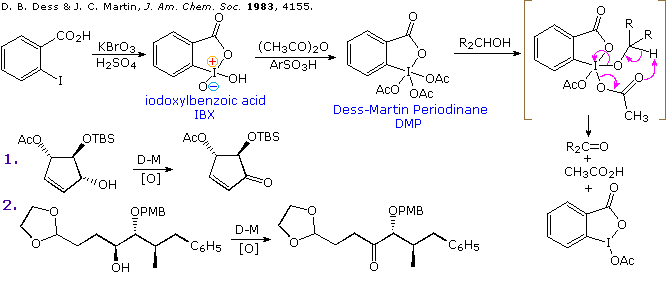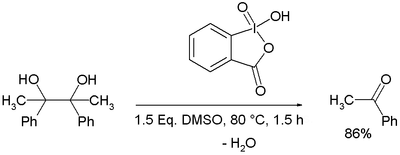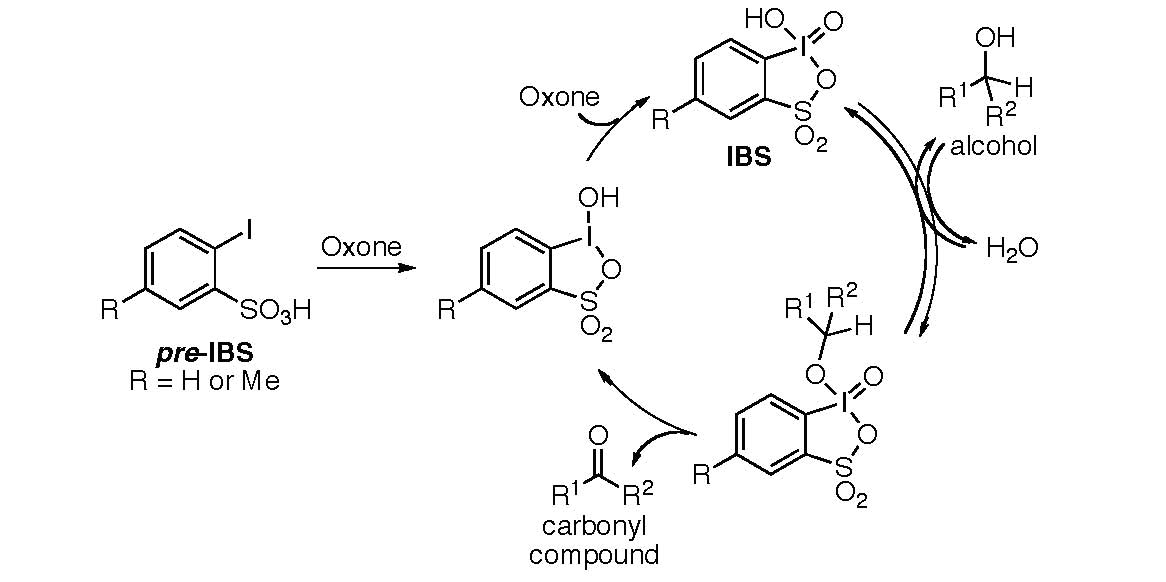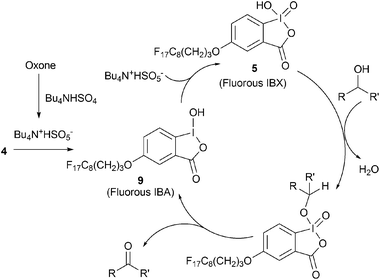
An effective and catalytic oxidation using recyclable fluorous IBX - Chemical Communications (RSC Publishing) DOI:10.1039/C0CC03149F

o-Iodoxybenzoic Acid (IBX) as a Viable Reagent in the Manipulation of Nitrogen- and Sulfur-Containing Substrates: Scope, Generality, and Mechanism of IBX-Mediated Amine Oxidations and Dithiane Deprotections | Journal of the American Chemical

IBX‐Mediated Dehydrogenation of Substituted β‐Oxonitriles - Klahn - 2014 - European Journal of Organic Chemistry - Wiley Online Library

New Reactions of IBX: Oxidation of Nitrogen‐ and Sulfur‐Containing Substrates To Afford Useful Synthetic Intermediates - Nicolaou - 2003 - Angewandte Chemie - Wiley Online Library
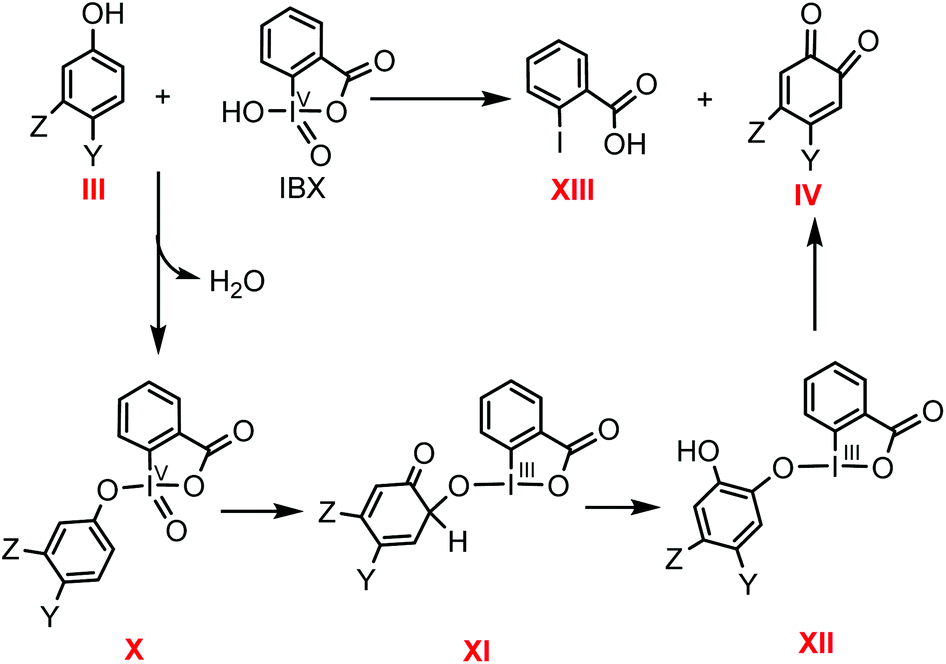
Mechanistic investigation into phenol oxidation by IBX elucidated by DFT calculations - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C9OB02650A

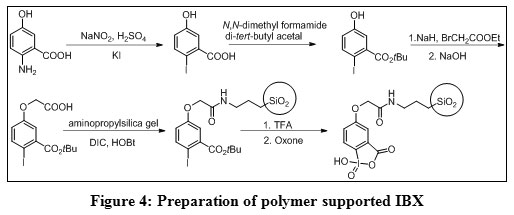
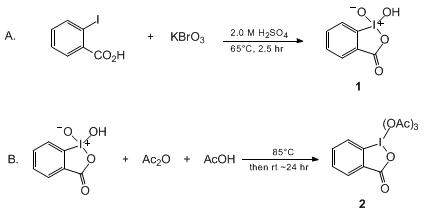
2-Iodoxybenzoic Acid: An Oxidant for Functional Group Transformations : Oriental Journal of Chemistry

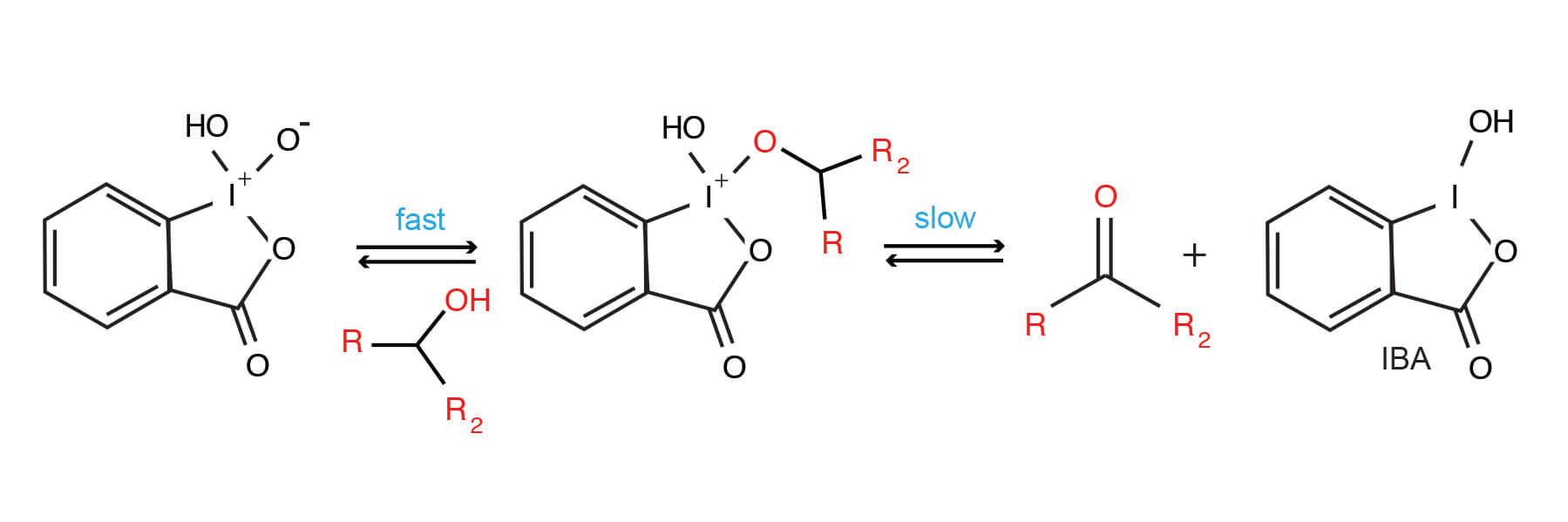
Scheme 4. General mechanism of oxidation of primary alcohols with IBX. | Download Scientific Diagram

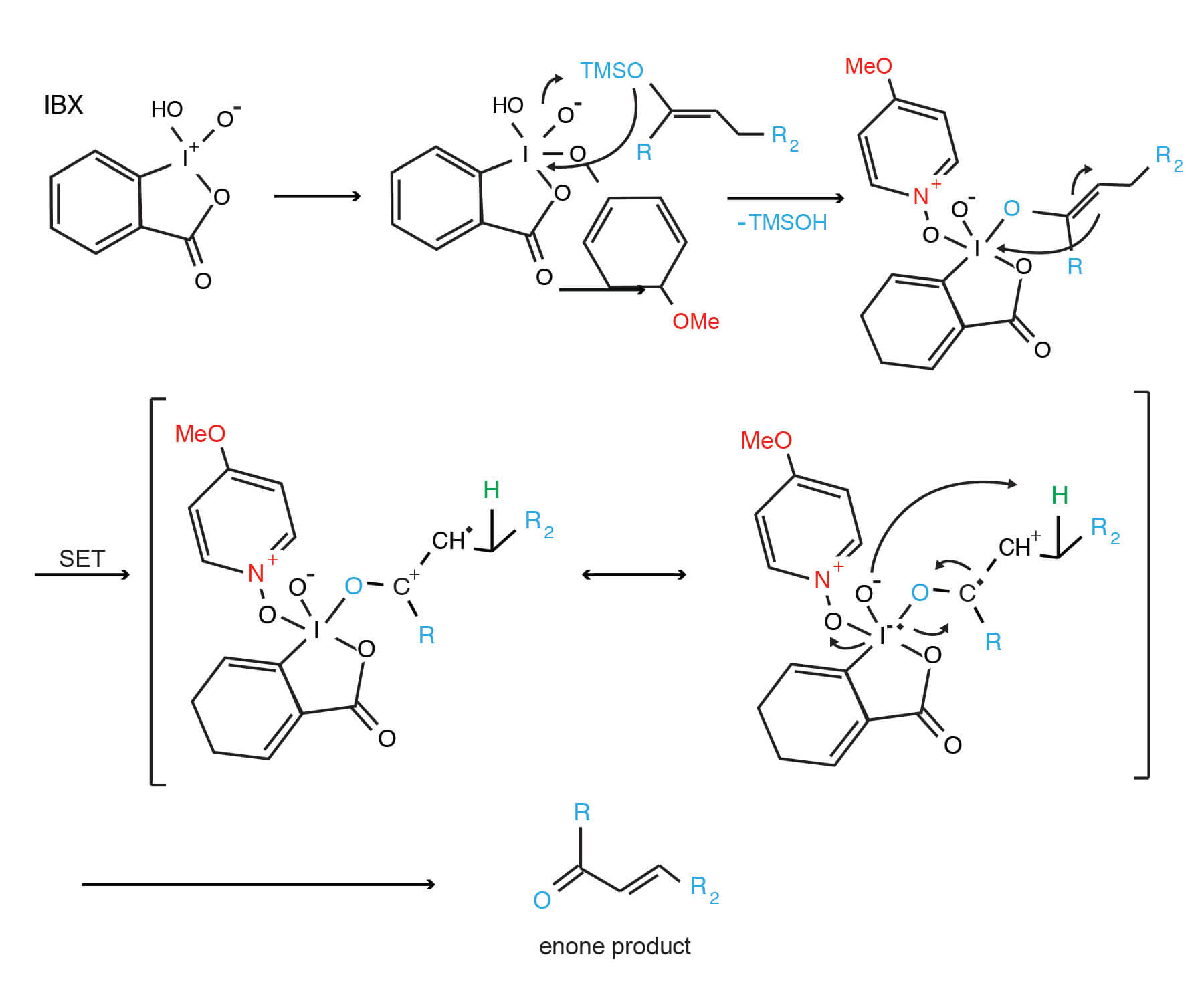
Oxidation of Silyl Enol Ethers by Using IBX and IBX⋅N‐Oxide Complexes: A Mild and Selective Reaction for the Synthesis of Enones - Nicolaou - 2002 - Angewandte Chemie International Edition - Wiley Online Library

Figure 3 from Iodine(V) reagents in organic synthesis. Part 4. o-Iodoxybenzoic acid as a chemospecific tool for single electron transfer-based oxidation processes. | Semantic Scholar