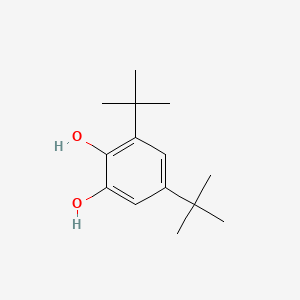
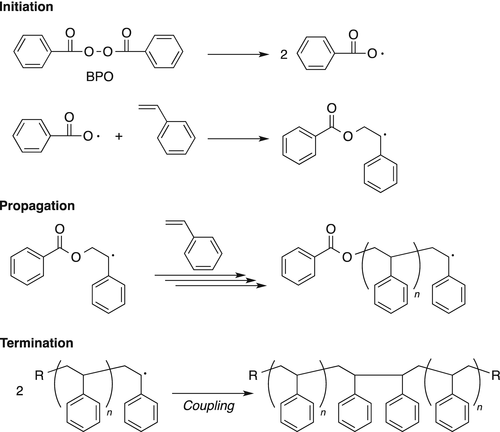
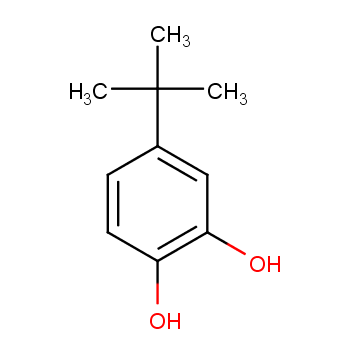
Processes | Free Full-Text | A Theoretical and Experimental Study for Screening Inhibitors for Styrene Polymerization
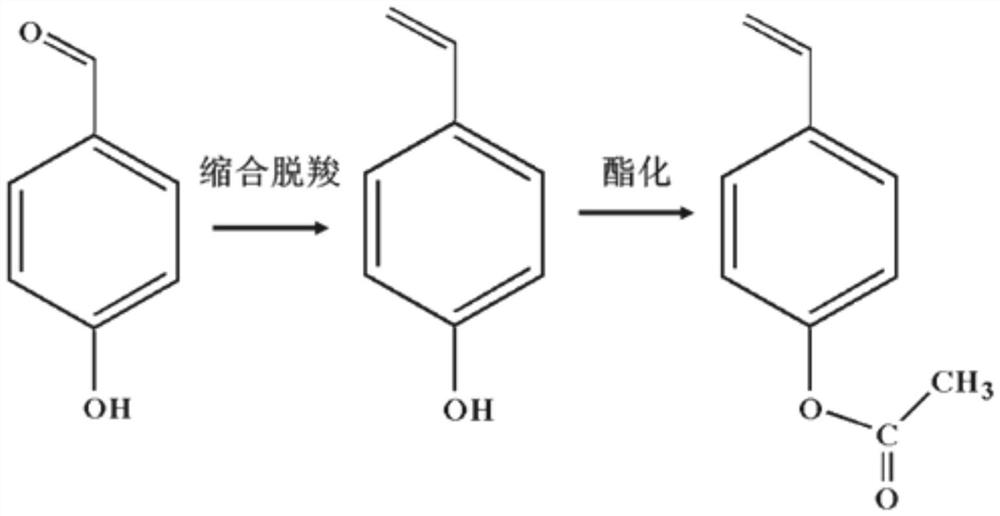
Method for preparing p-acetoxystyrene by one-pot method - Eureka | Patsnap develop intelligence library
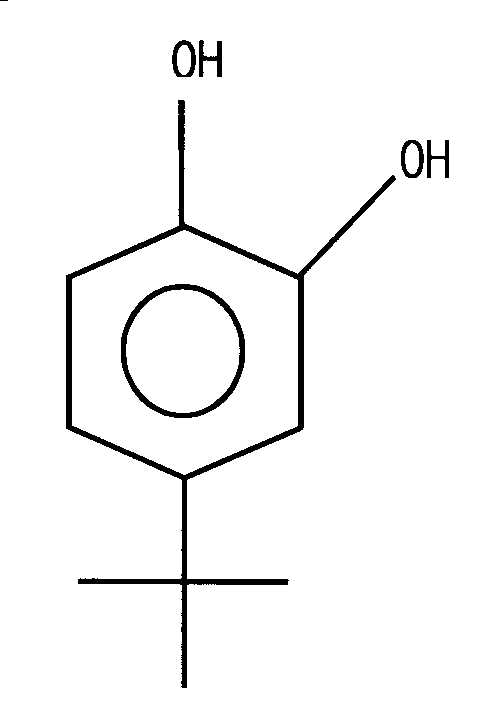
KR100676996B1 - Polymerization inhibitor for 1,3-butadiene and a method of inhibiting polymerization of 1,3-butadiene by imputing thereof - Google Patents

Peroxy Radical Activated Addition of tert-Butylcatechol to 2,6-Di-tert-butyl-7-Substituted Quinone Methide Polymerization Retarders | Organic Process Research & Development
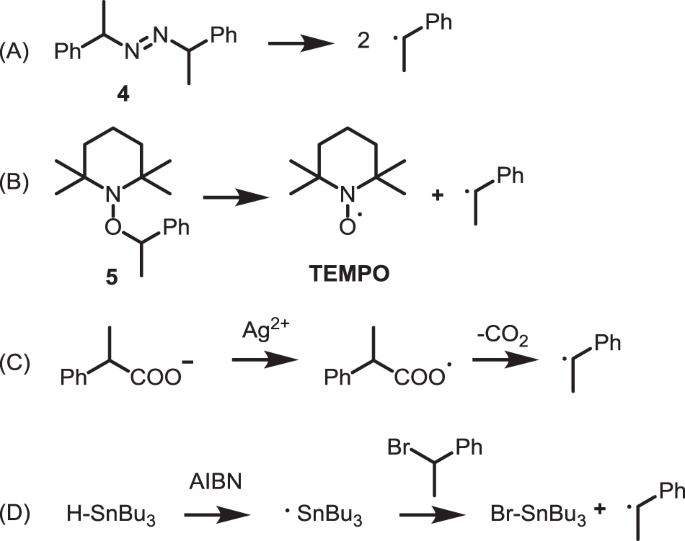
Polymerization inhibition mechanism of 1,4-naphthoquinone by experimentation and DFT calculations | Polymer Journal

Peroxy Radical Activated Addition of tert-Butylcatechol to 2,6-Di-tert-butyl-7-Substituted Quinone Methide Polymerization Retarders | Organic Process Research & Development

The influence of 4‐tert‐butylcatechol on the emulsion polymerization of styrene - Kemmere - 1999 - Journal of Applied Polymer Science - Wiley Online Library
![Polymerization Inhibitors | [Synthesis & Materials]Products | Laboratory Chemicals-FUJIFILM Wako Chemicals U.S.A. Corporation Polymerization Inhibitors | [Synthesis & Materials]Products | Laboratory Chemicals-FUJIFILM Wako Chemicals U.S.A. Corporation](https://labchem-wako.fujifilm.com/us/category/images/00209_img01.jpg)
Polymerization Inhibitors | [Synthesis & Materials]Products | Laboratory Chemicals-FUJIFILM Wako Chemicals U.S.A. Corporation

The synthesis, characterization and catecholase activity of dinuclear cobalt( ii / iii ) complexes of an O-donor rich Schiff base ligand - New Journal of Chemistry (RSC Publishing) DOI:10.1039/C4NJ00715H
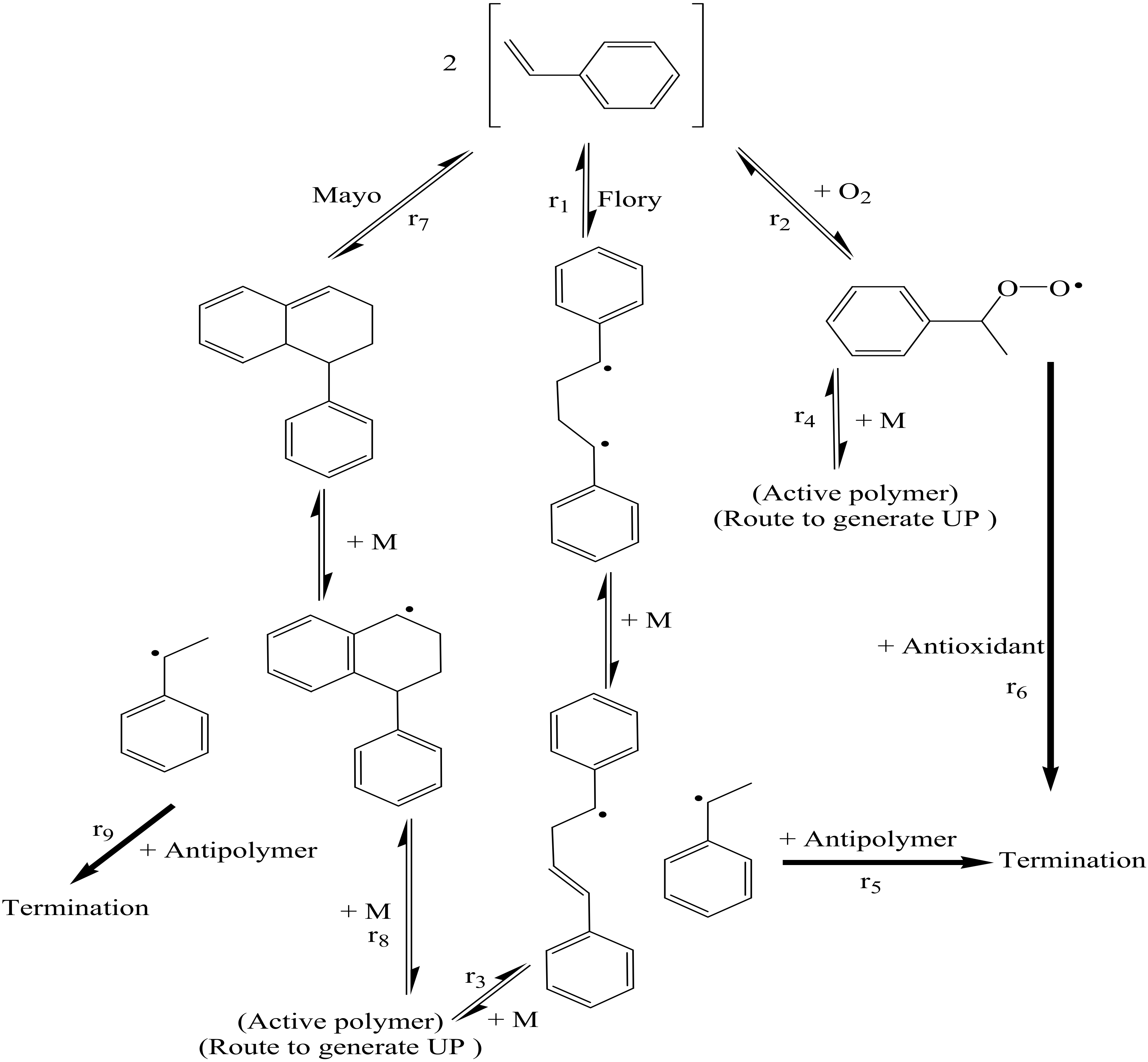
Processes | Free Full-Text | A Theoretical and Experimental Study for Screening Inhibitors for Styrene Polymerization

Processes | Free Full-Text | A Theoretical and Experimental Study for Screening Inhibitors for Styrene Polymerization